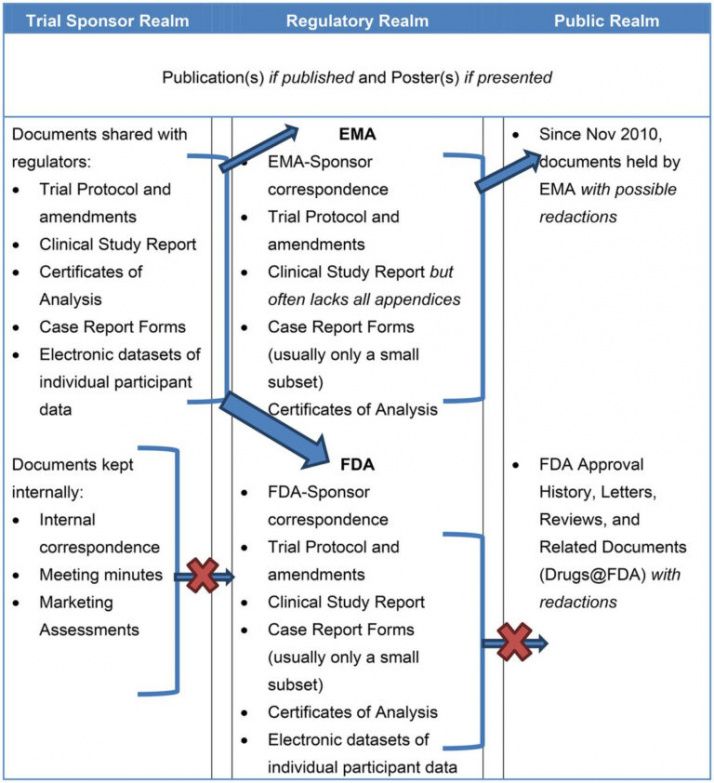
Clinical Study Report Template
Clinical Study Report Template - This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. Read together with international conference on. As such, this csr template is the foundation for an “integrated” full report of any study with a therapeutic, prophylactic, or diagnostic agent (i.e., drug or treatment) conducted in patients or.
As such, this csr template is the foundation for an “integrated” full report of any study with a therapeutic, prophylactic, or diagnostic agent (i.e., drug or treatment) conducted in patients or. Read together with international conference on. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or.
Read together with international conference on. As such, this csr template is the foundation for an “integrated” full report of any study with a therapeutic, prophylactic, or diagnostic agent (i.e., drug or treatment) conducted in patients or. The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions.
Speech And Language Report Template
As such, this csr template is the foundation for an “integrated” full report of any study with a therapeutic, prophylactic, or diagnostic agent (i.e., drug or treatment) conducted in patients or. Read together with international conference on. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions..
Give a report of a clinical case with this free medical presentation
The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. As such, this csr template is the.
Clinical Study Report Template PDF Sample Stableshvf
This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. As such, this csr template is the foundation for an “integrated” full report of any study with a therapeutic, prophylactic, or diagnostic agent (i.e., drug or treatment) conducted in patients or. Read together with international conference on..
Pin on Report Template
The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. Read together with international conference on. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. As.
Clinical Study Report (CSR) Template Clinical Study Templates
The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. Read together with international conference on. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. As.
(PDF) Clinical Study Report (CSR) Template DOKUMEN.TIPS
The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. Read together with international conference on. As such, this csr template is the foundation for an “integrated” full report of any study with a therapeutic, prophylactic, or diagnostic agent (i.e.,.
International Journal of Clinical Case Studies and Reports Template
The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. Read together with international conference on. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. As.
The enchanting The Basics Of Clinical Trial Centralized Monitoring For
This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. As such, this csr template is the.
Clinical Trial Report Template
The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. As such, this csr template is the foundation for an “integrated” full report of any study with a therapeutic, prophylactic, or diagnostic agent (i.e., drug or treatment) conducted in patients.
Medical Device Clinical Investigation Report
The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. Read together with international conference on. This document aims to allow the compilation of a single core clinical study report acceptable to all regulatory authorities of the ich regions. As.
This Document Aims To Allow The Compilation Of A Single Core Clinical Study Report Acceptable To All Regulatory Authorities Of The Ich Regions.
As such, this csr template is the foundation for an “integrated” full report of any study with a therapeutic, prophylactic, or diagnostic agent (i.e., drug or treatment) conducted in patients or. The clinical study report described in this guideline is an integrated full report of an individual study of any therapeutic, prophylactic or diagnostic agent (referred to herein as drug or. Read together with international conference on.