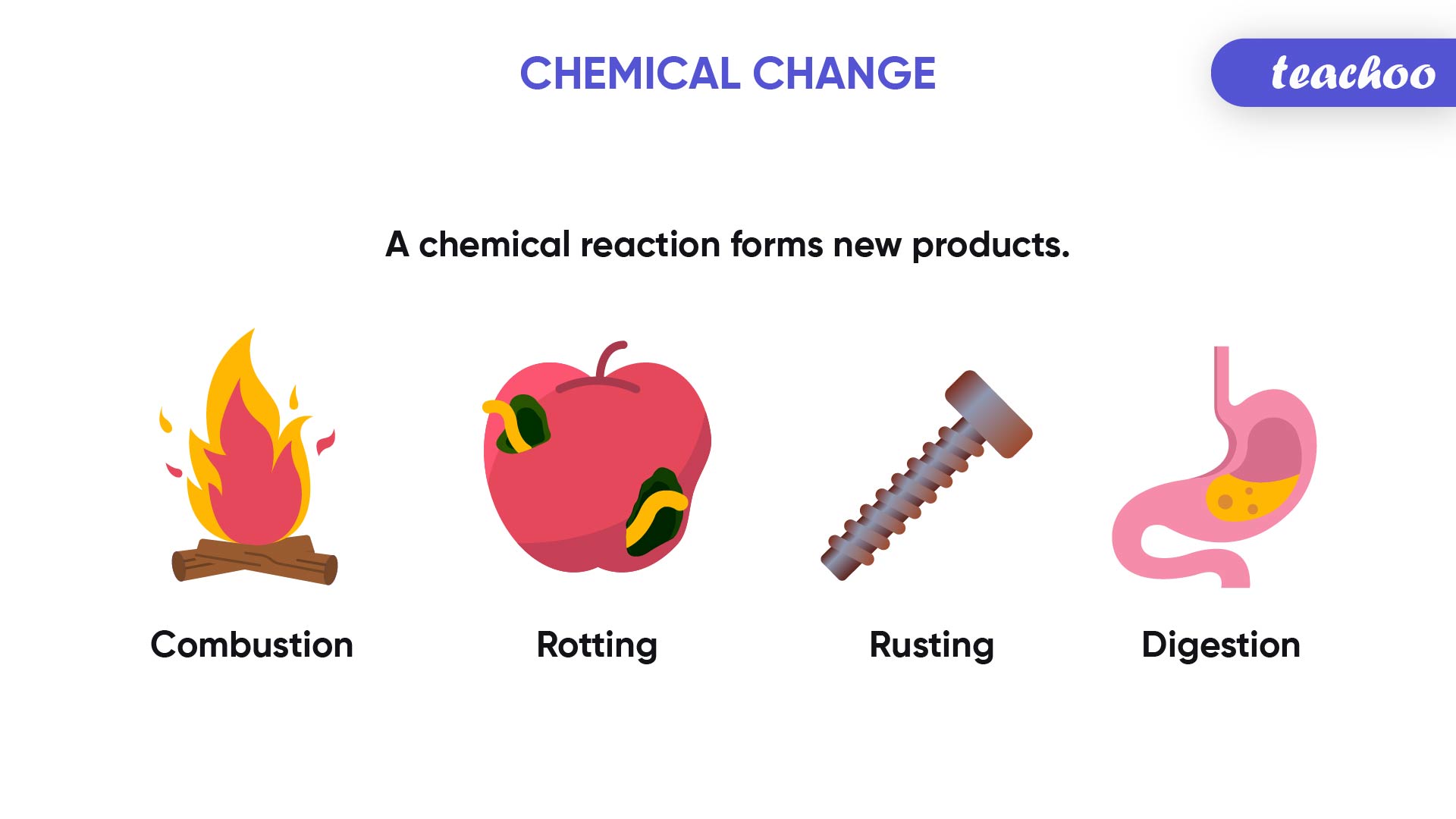
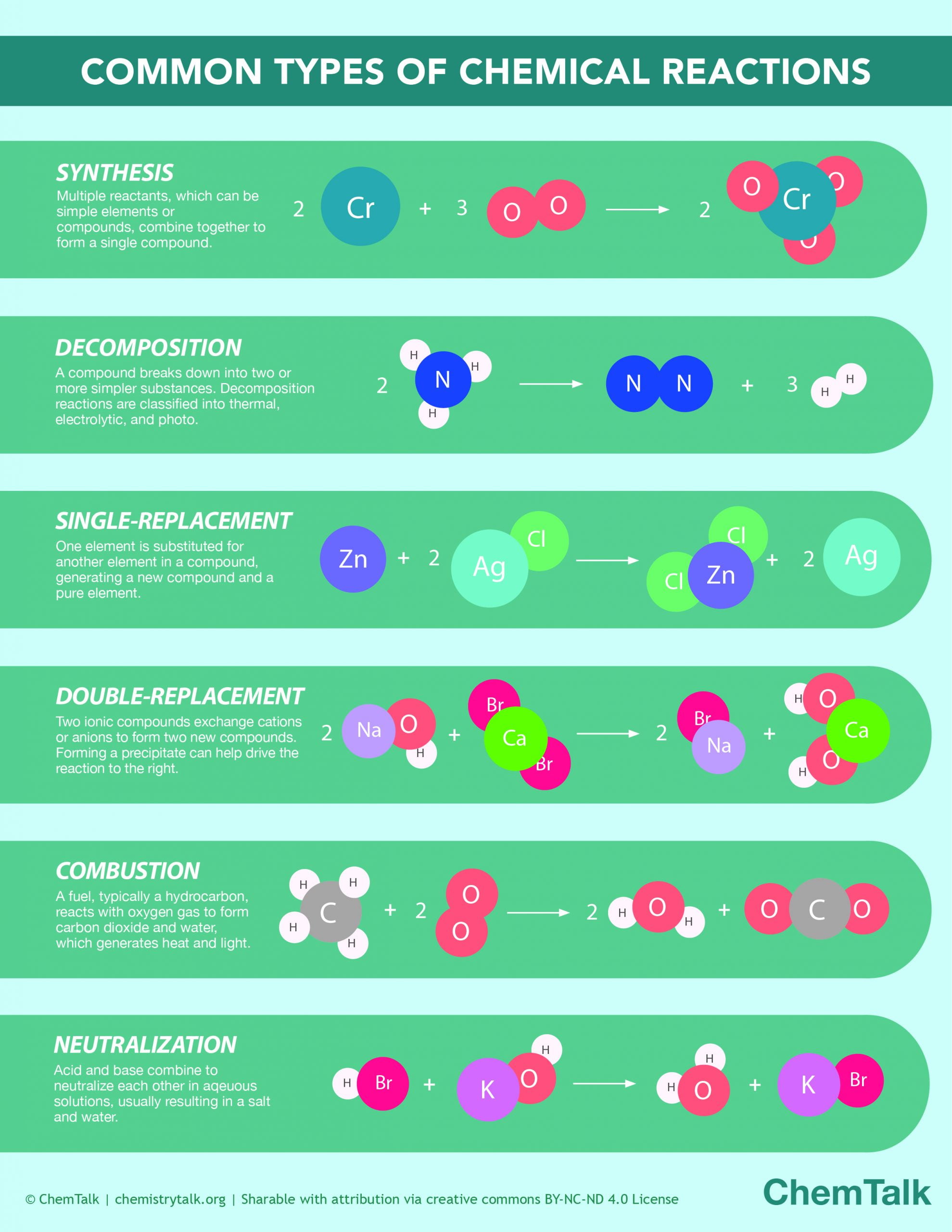
In Every Chemical Reaction What Is Conserved
In Every Chemical Reaction What Is Conserved - The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass of the reactants. Let’s analyze a few rows of the above table, beginning with row a. This is conserved in every ordinary chemical reaction. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Picture the reactants n 2 and h 2 as being the initial. The reactant that determines the amount of product that can be formed in a reaction.
This is conserved in every ordinary chemical reaction. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Picture the reactants n 2 and h 2 as being the initial. The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass of the reactants. Let’s analyze a few rows of the above table, beginning with row a. The reactant that determines the amount of product that can be formed in a reaction.
The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass of the reactants. The reactant that determines the amount of product that can be formed in a reaction. Let’s analyze a few rows of the above table, beginning with row a. This is conserved in every ordinary chemical reaction. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Picture the reactants n 2 and h 2 as being the initial.
Stoichiometry moltomol ratios ppt download
The reactant that determines the amount of product that can be formed in a reaction. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. This is conserved in every ordinary chemical reaction. Picture the reactants n 2 and h 2 as being the initial. Let’s analyze a few rows.
Stoichiometry ICS III Week ppt download
Picture the reactants n 2 and h 2 as being the initial. The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass of the reactants. Let’s analyze a few rows of the above table, beginning with row a. This is conserved in every ordinary chemical reaction. The reactant.
Drill What is a chemical reaction What is
Picture the reactants n 2 and h 2 as being the initial. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass of the reactants. The reactant that.
SOLVED Which of the following are CONSERVED in every chemical reaction
The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass of the reactants. Let’s analyze a few rows of the above table, beginning with row a. The reactant that determines the amount of product that can be formed in a reaction. This is conserved in every ordinary chemical.
Review When converting FROM moles you MULTIPLY. ppt download
The reactant that determines the amount of product that can be formed in a reaction. This is conserved in every ordinary chemical reaction. Picture the reactants n 2 and h 2 as being the initial. Let’s analyze a few rows of the above table, beginning with row a. The law of conservation of matter says that in chemical reactions, the.
Stoichiometry Chapter ppt download
Picture the reactants n 2 and h 2 as being the initial. This is conserved in every ordinary chemical reaction. The reactant that determines the amount of product that can be formed in a reaction. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Let’s analyze a few rows.
What Is Conserved in Chemical Reactions? Sciencing
In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass of the reactants. Picture the reactants n 2 and h 2 as being the initial. Let’s analyze a.
Chemical Reaction Definition, Types and Examples Class 10 Science
Picture the reactants n 2 and h 2 as being the initial. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. Let’s analyze a few rows of the above table, beginning with row a. This is conserved in every ordinary chemical reaction. The law of conservation of matter says.
Stoichiometry Notes. In every chemical reaction, the mass and number of
Let’s analyze a few rows of the above table, beginning with row a. The reactant that determines the amount of product that can be formed in a reaction. This is conserved in every ordinary chemical reaction. Picture the reactants n 2 and h 2 as being the initial. In every chemical reaction, the same mass of matter must end up.
Chemical Reactions Infographic ChemTalk
Picture the reactants n 2 and h 2 as being the initial. The reactant that determines the amount of product that can be formed in a reaction. This is conserved in every ordinary chemical reaction. In every chemical reaction, the same mass of matter must end up in the products as started in the reactants. The law of conservation of.
In Every Chemical Reaction, The Same Mass Of Matter Must End Up In The Products As Started In The Reactants.
The reactant that determines the amount of product that can be formed in a reaction. Picture the reactants n 2 and h 2 as being the initial. This is conserved in every ordinary chemical reaction. The law of conservation of matter says that in chemical reactions, the total mass of the products must equal the total mass of the reactants.